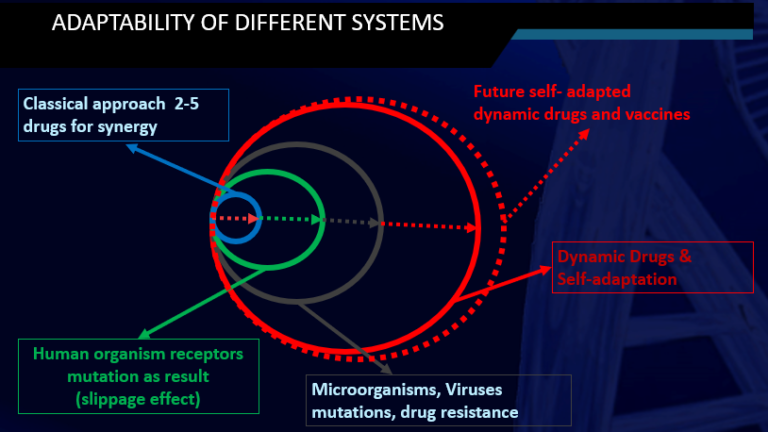
The figure presents a pie chart where the area reflects the degree
of freedom (variability, adaptability) of various “systems” in relation
to external conditions:
If we visualize the degree of variability of traditional drugs in
the human population as a blue circle, the adaptability of
microorganisms, fungi, and viruses overlaps this variability, prompting
pharmaceutical companies to develop new antibiotics.
These antibiotics re-enter the blue circle, yet microbes continue
to evade them. The green area illustrates the impact of this “slipping”
effect on the human body, where aging and cell renewal (the human body
is inherently dynamic) cause the drug’s variability area to overlap
over time. As a result of this slipping effect, cardiologists must
adjust the combinations of hypotensive medications for individuals
periodically. Given the redundancy of components in dynamic drugs with
self-adaptive capabilities, their levels of variability and
adaptability significantly surpass both the slipping effect and the
adaptive responses of microorganisms (depicted as red circles). This
enables dynamic drugs to effectively combat the resistance exhibited by
microorganisms and viruses.
An essential place in the modern structure of the pharmaceutical
market belongs to drugs: biotechnological products of various origins.
These drugs, such as recombinant insulins, interferons, interleukins,
erythropoietin, and so on, are essential to patients’ lives. At the
same time, statistical data on introducing these drugs on the market
and several low-molecular drugs demonstrate that they are
insignificantly more effective than a placebo. For example, the use of
beta-interferon (in the treatment of disseminated sclerosis) exceeds
the placebo in effectiveness by only 8 % (placebo: 30%;
beta-interferon: 38%). The situation is almost the same for
low-molecular drugs such as antihypertensives. The effectiveness of
amlodipine at the third stage of clinical tests was only 22% higher
than that of the placebo (amlodipine: 52%; placebo: 30%). The reasons a
drug might be ineffective or of little effect for 48% of patients have
not yet been determined. The most difficult to explain is the
ineffectiveness of drugs whose acceptors are cell receptors that have
long been the study subjects, namely, adreno-, choline-, and histamine
receptors. It remains a mystery why the same drug can be ineffective
for one group of patients while remaining practical for another. Due to
this little-studied peculiarity of the human organism, most
antihypertensive drugs are combined. It is especially necessary to
combine at least three drugs with different mechanisms of action but
the same result: antihypertensive or cytostatic. In the latter case,
the differences between various kinds of tumors are specially marked in
sensitivity and the individual peculiarities of a specific tumor and
host organism. Even polychemotherapy often turns out to be ineffective
in the treatment of patients with cancer. The FDA’s depressing
statistics on the third stage of clinical tests of drugs demonstrate
the low effectiveness of practically all medicinal drugs available on
the pharmaceutical market. The average efficacy of the most potent drug
(morphine) is 75%. In other cases, we observe intolerance and toxic
effects or an opposite reaction. Even in narcotic drug applications,
only 60% of the people who took them are observed to experience the
classical impact. The other group of people who took, for example,
cocaine suffered a severe headache and dizziness without any signs of
anesthesia. This kind of divergence of effects may be caused by
polymorphism of the receptor system within the human population.
Earlier, the structure of receptors was considered to be absolutely
conservative and invariable for a single, or sometimes several, animal
species. At present, more and more scientists tend to believe that
receptors differ in much the same way as human faces, even within a
single species. These differences are caused not only and not so much
by the change of the primary amino acid sequence of receptors’ protein
base but also by conformation changes of secondary and tertiary
structures. Although they are formally similar in primary structure and
molecular weight, different people’s receptors are actually different
combinations of protein isoforms. This is especially apparent in the
example of a major histocompatibility complex (MHC) antigen isoform
combination. The selection of this complex is vitally important to
organ transplantation processes. If thousands of variants and
combinations exist for the MHC system, why should the structures of an
organism’s other receptors be conservative within a species? Most
likely, similarly to MHC antigens, the tertiary structures of the
majority of cell receptors differ significantly by isoform profile
within a population. This hypothesis provides a good explanation for
drugs’ low levels of effectiveness. A conservative structure of a
classical drug (like one“key”) cannot match a specific receptor (“many
different, though similar, locks”) in all individuals of one species
equally and with equal affinity.

Illustration
1. Instead of one “key” for one “lock” (the principle of a classic
drug with a conservative structure), we propose a selection of
“skeleton keys”: a group of many similar molecules that “open” many
“locks” and adapt to the target. This
facilitates a practically 100% effectiveness rate and a maximally wide
spectrum of drug activity.
To increase drug effectiveness, it is necessary to change the
concept of drug development and approaches to this process. For
example, docking, one of the most effective methods of modern drug
design, uses a conservative sequence of one receptor. Sometimes, target
conformations are used in different solvents with further overlay of
conformations. Obtaining infinitely many receptors and finding ONE
inhibitor substance for all conformations will never be possible. One
of nature’s successful solutions is immunoglobulin system evolution,
which protects higher organisms from external aggressive factors, such
as viruses, microorganisms, and fungi. Practically, the same
immunoglobulin base (Fcareas and heavy chains) with a large number of
different variants on FAB – fragments being specific to their targets –
is quite a successful solution. In this situation, affinity to targets
(the level of correspondence of the “hand” to the “glove”) may vary
from 5% of IgM to 95% of IgG.
Illustration
2. There are five basic forms of immunoglobulins in order of their
synthesis and strengthening of affinity (relationship) and their
specificity to targets (the ability to selectively find and interact
with a target): IgM; IgA; IgE;
IgD;IgG
Sometimes one target antigen may cause the generation of several
hundred thousand variants of immunoglobulins with different monoclonal
specificity for different epitopes. This kind of polymorphism justifies
itself: the majority of the population survives infectious diseases. In
many cases, reconvalescent donor immunoglobulins are still the only
effective means of treating many diseases, such as SARS and Marburg
fever. To provide the maximum affinity for the maximum number of
people, it is necessary to have a mixture of millions of molecules that
are very similar but still different from each other in one vial. In
this case, we do not obtain one “key” but a whole bunch of keys. At
least one “key” from this “bunch” will match a specific patient and
his/her original receptor. Suppose it is impracticable under modern
conditions to synthesize a specific inhibitor for a specific patient.
In that case, the only option is to produce millions of inhibitor
isoforms in one mole of a substance. In our opinion, one of the most
reasonable methods of solving the problem of low drug effectiveness is
to obtain precision partially chemically modified recombinant
biotechnological preparations: biopolymers (proteins, polysaccharides,
polynucleotides, tannins, self-organized structures/phospholipids,
etc.). The usage of this technology will bring pharmaceutical science
to a level of intensive development, make molecular modeling methods
much more straightforward, and increase the probability of a practical
means of introducing therapeutic drugs. The effectiveness of this
design method (drag design) for medications is confirmed by the
effectiveness of the antiviral veterinary drug SPRO-2018, which was
designed and implemented based on self-organizing quasi-life systems.
This drug is effective in treating nearly all existing viral infections
in animals, even at very severe stages of the disease, with quick
therapeutic manifestation. This drug has received permission for
production and application as a veterinary medicine. A second drug,
FRNA-211, is a self-organizing quasi-life system based on modified RNA.
It has the properties of microRNA and has shown significant cytostatic
properties in more than 50 cell cultures and in three animal models.
Research in the area of substituting dynamic medications for
conservative structures continues.
Presentation for Dynamic Drugs: Dynamic
