MoLViRx
APPROACH
One of the ways to create new antiviral drugs that are effective in treating diseases caused by RNA viruses is the selection of RNA-dependent RNA polymerase (RdRp) inhibitors using molecular modeling methods [16–18]. Docking led to the development of molnupiravir, the only drug effective in mitigating the effects of SARS-CoV-2 [19,20]. Although the drug alleviates symptoms, it does not significantly reduce patient mortality from complications [21]. In addition, it is effective only in the initial stages of infection. It was previously discovered that cobolamines in high doses can inhibit the replication of several viruses in cell cultures [6,22]. The mechanism of action of cobalamin was determined to be inhibition of RdRp through simultaneous interaction with specific aptamer fragments of viral RNA and the peptide fragment of RdRp. At the same time, the dose of cyanocobalamin was quite high and exceeded what was permissible for use in vivo. We hypothesized that it is possible to increase the affinity of cobalamin for the binding sites of RNA aptamers and RdRp by carboxylation of ribose residues. This may increase the affinity of such a product for other binding sites and reduce the effective dose of the drug [23].
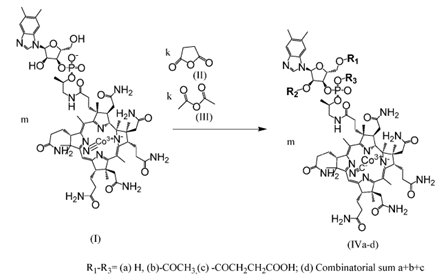
As can be seen from Figure 1, the carboxylation reaction results in a mixture of derivatives substituted at the ribose residue with different combinations and substitutions. A total of 8 different cyanocobalamin derivatives are formed. Each component of this mixture synergistically enhances the effects of the others because it has different tropism to different points of interaction with the RNA-aptamer (Fig. 2-3).
Preliminary results
In silico study
Based on the results of molecular modeling (Yasara, Autodock Vina, aptamer 1et4, cyanocobalamin alone (Fig. 2 and dynamic structure MoLviRx from 8 derivatives Fig. 3), we have shown that the degree of affinity of B12 to RNA aptamer fragments of viral RNA and the protein component of the SARS-CoV-2 RdRp can be increased by several times if the ribose fragment of cyanocobalamin is combinatorial carboxylated with several modifiers simultaneously. Such structures containing a mixture of combinatorial derivatives with the same core but different dicarboxylic acid residues in the ribose fragment are called dynamic. They can form supramolecular structures with the revert RdRp and viral RNA complex and integrate into various regions of this structure, different from those unmodified B12 binds.

As can be seen from Figure 1, cyanocobalamin has 3 binding points to the aptamer on its surface, while the dynamic structure MoLviRx (Figure 2) binds to the aptamer at 7 points. Thus, MoLviRx's multiple targeting makes it possible to reduce its effective dose and reduce the likelihood of the virus adapting to the drug.
We have successfully synthesized a novel structure - dynamic carboxylated cyanocobalamin, which includes a mixture of 8 derivatives (MoLviRx ). Combinatorial synthesis involved using non-equivalent amounts of two modifiers simultaneously - maleic and succinic anhydride. This innovative approach allows all 8 derivatives to function as a combination chemotherapy, targeting different regions of viral RNA aptamers and blocking various RNA replication mechanisms. This unique strategy eliminates the risk of drug resistance development. Initial in vitro studies have revealed a broad spectrum of action of MoLviRx on viruses such as influenza (H1N1-09), New Castle disease virus (live vaccinia virus Merck), and coronavirus (str.D52) at doses 100 times lower (8-10 mcM/mL) than the original Cyanocobalamin (900-1200 mcM/mL). A culture virus titer decrease was observed from 8 lg to 2 lg. The MoLviRx product we developed is protected by two valid patents US11160878B1 and US11191806B2. In addition to its higher antiviral activity, MoLviRx has shown significant nephroprotective properties and a higher affinity for renal megalin than native B12.
Next, we provide an example of studies of the developed product's antiviral activity on the TGP pig gastroenteritis coronavirus model.

Antiviral activity in vitro to coronavirus str.D52
The purpose of the study is to determine the virucidal activity of the MoLviRx substance in a model of coronavirus transmissible gastroenteritis of pigs (TGP).
Materials and methods
Virus: TGEV - the etiological agent of transmissible gastroenteritis of pigs (TGP) - a highly contagious intestinal disease of pigs (coronavirus strain str.D52) [24]. D52-5 (BRE79) - is a highly pathogenic virus for pigs of all ages at 5 passages in transplanted monolayer culture of testicular cells of piglets ST. The tropism of the virus in the gastrointestinal tract and respiratory tract is shown. Dr. Hubert Laude provided the strain from the Laboratory of Molecular Virology and Immunology of the INRA Center for Biotechnology in Jouy-en-Josas (France). Cell culture: CHEB - is a transplanted culture of kidney cells of a pig embryo [25]; substance: MOLVIRX - dynamic derivatives of cyanocobalamin 1% solution.
Determination of antiviral action. A str.D52 with an infectious titer 8,5 lg ID50 was used. These studies aim to identify the antiviral effect of the MoLviRx substance. The viral suspension, at a dose of TCD50/ml, was incubated with a cell culture CHEB for 4 hours. After that, 0.001% and 0.0001% (up to the final concentration in the medium) MoLviRx solution was added to the culture, and the cells were allowed to continue incubating. The infectious titer (viral RNA copies in terms of titer) of the virus was then determined the next day by RT-PCR. A decrease in the infectious titer of the virus by 1.5-2.0 lgTCD50 compared to the control indicates the severity of the antiviral effect.
Detection of RNA of transmissible gastroenteritis virus of pigs str.D52 by reverse polymerase chain reaction (RT-PCR). RNA isolation was performed using a set of "Fish-sorb" following the manufacturer's instructions (AmpliSens). The reverse transcription reaction was performed using a kit called "RevertAidTM H Minus First Strand DNA Synthesis Kit" according to the manufacturer's instructions (ThermoScientific) [26]. For PCR were used gene-specific nucleoprotein oligonucleotide primers of the following sequence: direct Uni_1 (5'-TGCACTGATCAATGTGCTAG-3) and reversed Uni 2 (5' TGAAAACACTGTGGCACCCТТ-3") [27]. A fragment amplified by size 309 P.M. M-marker "100 bpPlus DNA Ladder" ("ThermoFisherScientific") [28]. TGP virus from different cultures was propagated in CHEB culture, and the amplification products were by distributing DNA fragments in 1.5% aerosol gel.
Statistical processing of research results. The digital material presented in the work is processed statistically. Statistical evaluation of the significance levels of differences in the obtained figures was performed using Student's t-test using Microsoft Excel and Microcal Origin. Differences at p <0.05 were considered authentic.
Research results
The results showed that the MoLviRx substance inhibited the reproduction of extracellular coronavirus TGP by 5.0 lg at dose 0,0001% min exposure and 6.0 lg at dose 0,001%.
Conclusions
MoLviRx's antiviral activity is dose-dependent. Research has determined that it is an effective antiviral substance and can be used to develop new anticoronaviral drugs.
Team Expertise: Our team uniquely integrates the Theory of Inventive Problem Solving (TRIZ) into pharmaceutical development and includes Ph.D. scientists with diverse educational and practical backgrounds. We are composed of experts in pharmacology, microbiology, and drug design and have recently secured 10 new patents in drug design, including three in antivirals, bringing our total to over 220 granted patents, with several industry-implemented innovations. Our principal investigator and consultant are the authors of the proposal’s granted patents and peer-reviewed published articles. Key personnel include a clinician experienced in anticancer trials and multicenter studies. Our diverse expertise and systems approach position us to tackle complex challenges effectively.
One of the ways to create new antiviral drugs that are effective in treating diseases caused by RNA viruses is the selection of RNA-dependent RNA polymerase (RdRp) inhibitors using molecular modeling methods [16–18]. Docking led to the development of molnupiravir, the only drug effective in mitigating the effects of SARS-CoV-2 [19,20]. Although the drug alleviates symptoms, it does not significantly reduce patient mortality from complications [21]. In addition, it is effective only in the initial stages of infection. It was previously discovered that cobolamines in high doses can inhibit the replication of several viruses in cell cultures [6,22]. The mechanism of action of cobalamin was determined to be inhibition of RdRp through simultaneous interaction with specific aptamer fragments of viral RNA and the peptide fragment of RdRp. At the same time, the dose of cyanocobalamin was quite high and exceeded what was permissible for use in vivo. We hypothesized that it is possible to increase the affinity of cobalamin for the binding sites of RNA aptamers and RdRp by carboxylation of ribose residues. This may increase the affinity of such a product for other binding sites and reduce the effective dose of the drug [23].

Figure 1.
An example of the combinatorial synthesis of one of the MoLviRx variants (modifiers succinic and acetic
anhydrides)
As can be seen from Figure 1, the carboxylation reaction results in a mixture of derivatives substituted at the ribose residue with different combinations and substitutions. A total of 8 different cyanocobalamin derivatives are formed. Each component of this mixture synergistically enhances the effects of the others because it has different tropism to different points of interaction with the RNA-aptamer (Fig. 2-3).
Preliminary results
In silico study
Based on the results of molecular modeling (Yasara, Autodock Vina, aptamer 1et4, cyanocobalamin alone (Fig. 2 and dynamic structure MoLviRx from 8 derivatives Fig. 3), we have shown that the degree of affinity of B12 to RNA aptamer fragments of viral RNA and the protein component of the SARS-CoV-2 RdRp can be increased by several times if the ribose fragment of cyanocobalamin is combinatorial carboxylated with several modifiers simultaneously. Such structures containing a mixture of combinatorial derivatives with the same core but different dicarboxylic acid residues in the ribose fragment are called dynamic. They can form supramolecular structures with the revert RdRp and viral RNA complex and integrate into various regions of this structure, different from those unmodified B12 binds.

Figure 2. Yasara, Autodock Vina, multiligand docking,
aptamer 1et4, cyanocobalamin alone. 3 binding points.
As can be seen from Figure 1, cyanocobalamin has 3 binding points to the aptamer on its surface, while the dynamic structure MoLviRx (Figure 2) binds to the aptamer at 7 points. Thus, MoLviRx's multiple targeting makes it possible to reduce its effective dose and reduce the likelihood of the virus adapting to the drug.
We have successfully synthesized a novel structure - dynamic carboxylated cyanocobalamin, which includes a mixture of 8 derivatives (MoLviRx ). Combinatorial synthesis involved using non-equivalent amounts of two modifiers simultaneously - maleic and succinic anhydride. This innovative approach allows all 8 derivatives to function as a combination chemotherapy, targeting different regions of viral RNA aptamers and blocking various RNA replication mechanisms. This unique strategy eliminates the risk of drug resistance development. Initial in vitro studies have revealed a broad spectrum of action of MoLviRx on viruses such as influenza (H1N1-09), New Castle disease virus (live vaccinia virus Merck), and coronavirus (str.D52) at doses 100 times lower (8-10 mcM/mL) than the original Cyanocobalamin (900-1200 mcM/mL). A culture virus titer decrease was observed from 8 lg to 2 lg. The MoLviRx product we developed is protected by two valid patents US11160878B1 and US11191806B2. In addition to its higher antiviral activity, MoLviRx has shown significant nephroprotective properties and a higher affinity for renal megalin than native B12.
Next, we provide an example of studies of the developed product's antiviral activity on the TGP pig gastroenteritis coronavirus model.

Figure 3. Yasara, Autodock Vina, multi-ligand docking,
aptamer 1et4, MoLviRx 7 binding points
Antiviral activity in vitro to coronavirus str.D52
The purpose of the study is to determine the virucidal activity of the MoLviRx substance in a model of coronavirus transmissible gastroenteritis of pigs (TGP).
Materials and methods
Virus: TGEV - the etiological agent of transmissible gastroenteritis of pigs (TGP) - a highly contagious intestinal disease of pigs (coronavirus strain str.D52) [24]. D52-5 (BRE79) - is a highly pathogenic virus for pigs of all ages at 5 passages in transplanted monolayer culture of testicular cells of piglets ST. The tropism of the virus in the gastrointestinal tract and respiratory tract is shown. Dr. Hubert Laude provided the strain from the Laboratory of Molecular Virology and Immunology of the INRA Center for Biotechnology in Jouy-en-Josas (France). Cell culture: CHEB - is a transplanted culture of kidney cells of a pig embryo [25]; substance: MOLVIRX - dynamic derivatives of cyanocobalamin 1% solution.
Determination of antiviral action. A str.D52 with an infectious titer 8,5 lg ID50 was used. These studies aim to identify the antiviral effect of the MoLviRx substance. The viral suspension, at a dose of TCD50/ml, was incubated with a cell culture CHEB for 4 hours. After that, 0.001% and 0.0001% (up to the final concentration in the medium) MoLviRx solution was added to the culture, and the cells were allowed to continue incubating. The infectious titer (viral RNA copies in terms of titer) of the virus was then determined the next day by RT-PCR. A decrease in the infectious titer of the virus by 1.5-2.0 lgTCD50 compared to the control indicates the severity of the antiviral effect.
Detection of RNA of transmissible gastroenteritis virus of pigs str.D52 by reverse polymerase chain reaction (RT-PCR). RNA isolation was performed using a set of "Fish-sorb" following the manufacturer's instructions (AmpliSens). The reverse transcription reaction was performed using a kit called "RevertAidTM H Minus First Strand DNA Synthesis Kit" according to the manufacturer's instructions (ThermoScientific) [26]. For PCR were used gene-specific nucleoprotein oligonucleotide primers of the following sequence: direct Uni_1 (5'-TGCACTGATCAATGTGCTAG-3) and reversed Uni 2 (5' TGAAAACACTGTGGCACCCТТ-3") [27]. A fragment amplified by size 309 P.M. M-marker "100 bpPlus DNA Ladder" ("ThermoFisherScientific") [28]. TGP virus from different cultures was propagated in CHEB culture, and the amplification products were by distributing DNA fragments in 1.5% aerosol gel.
Statistical processing of research results. The digital material presented in the work is processed statistically. Statistical evaluation of the significance levels of differences in the obtained figures was performed using Student's t-test using Microsoft Excel and Microcal Origin. Differences at p <0.05 were considered authentic.
Research results
The results showed that the MoLviRx substance inhibited the reproduction of extracellular coronavirus TGP by 5.0 lg at dose 0,0001% min exposure and 6.0 lg at dose 0,001%.
Conclusions
MoLviRx's antiviral activity is dose-dependent. Research has determined that it is an effective antiviral substance and can be used to develop new anticoronaviral drugs.
Team Expertise: Our team uniquely integrates the Theory of Inventive Problem Solving (TRIZ) into pharmaceutical development and includes Ph.D. scientists with diverse educational and practical backgrounds. We are composed of experts in pharmacology, microbiology, and drug design and have recently secured 10 new patents in drug design, including three in antivirals, bringing our total to over 220 granted patents, with several industry-implemented innovations. Our principal investigator and consultant are the authors of the proposal’s granted patents and peer-reviewed published articles. Key personnel include a clinician experienced in anticancer trials and multicenter studies. Our diverse expertise and systems approach position us to tackle complex challenges effectively.
