APPROACH
One promising direction in cancer treatment is the development of gene
therapy based on antisense oligonucleotides. This approach, mainly
using complementary polynucleotides to inactivate genes, holds great
promise in the fight against cancer. However, cancer gene therapy faces
significant challenges: the destruction of synthetic complementary
nucleotides by blood nucleases, limited penetration into cells, and
sensitivity to cell genome repair systems. These hurdles require
immediate attention and further research. According to the principles
of Systems Evolution, all man-made systems are progressing towards
miniaturization. What was once large is now microscopic – from a
considerable radio lamp to a microprocessor and from robots to
nanorobots. These nanorobots are designed to mimic the adaptive
capabilities of living organisms, responding to environmental
conditions within the body. Specifically, our technology draws
inspiration from viral behavior – the self-assembly of proteins and
polynucleotides into complex virions. We aim to develop a new class of
adaptive, multi-functional drug delivery systems by applying this
principle to therapeutic nanostructures.
While
one molecule can perform a single function, complex nanostructures
based on modified RNA and peptides can carry out multiple sequential
operations. They can also self-organize and self-assemble into more
intricate and hybrid structures with entirely different properties than
the original components. This behavior resembles viruses, which
self-assemble from a mix of proteins and polynucleotides into complex
virions. Our developed nanorobots are supramolecular structures based
on peptides or quasi-living nanostructures. Living organisms can adapt
to their surroundings. We have proposed structures based on
antisense-oligoRNA, which can self-assemble on cellular RNA (such as
tRNA, mRNA, and rRNA) and inhibit their functions. Different approaches
are taken to designing antisense oligonucleotides. However, the
principle of their interaction with targets remains the same: the
formation of hydrogen bonds between complementary nucleotides with
increased resistance to nucleases. It’s important to note that the
principle of gene inactivation through their complementary interaction
with antisense nucleotides remains consistent – the formation of
hydrogen bonds. These are also known as complementary miRNAs, which
selectively block the synthesis of specific proteins in the cell. It is
well known that many adenocarcinomas capture oligonucleotides and
nanoparticles through pinocytosis, while healthy cells cannot capture
small oligonucleotides and liposomes. This ensures selective
accumulation of the proposed protected oligonucleotides (antisense
oligoRNA – MoLRx) in cancer cells and the absence of toxicity of the
MoLRx composition. To obtain MoLRx, we used antisense oligomeric
fragments of RNA – recognized by ribosomes. This antisense oligo-RNA
gave the property of complementarity and protection against nucleases
by simultaneous combinatorial acylation without subsequent separation
of the combinatorial library into individual compounds. Selective
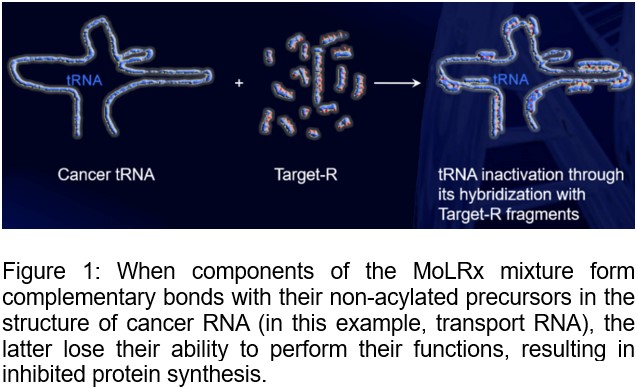
accumulation of the MoLRx in the cancer cell leads to cancer cell
hybridization with complementary targets in the cancer cell’s tRNAs,
mRNAs, and rRNAs (Figure 1). This results in a gradual halt in protein
synthesis due to the blockade of protein synthesis in incorporating
amino acids into the polypeptide chain. The action of MoLRx is based on
the induction of apoptosis through the termination of protein
synthesis. We designed antisense oligo RNAs (MoLRx), consisting of
thousands of fragments of different sizes (up to 30 nucleotides) and
sequences, which act similarly to polychemotherapy. After
carboxylation, these MoLRx exhibited the desired antisense properties
and could offer some benefits of polychemotherapy. Our research on
MoLRx aims to overcome multidrug resistance, potentially extending
patient survival and reducing mortality associated with MDR cancers.
The oligomeric fragments of RNA contain numerous adenine and guanine
groups that can be modified. A wide variety of derivatives with
different lengths and charges are created through combinatorial
synthesis. Each component in this mixture pairs with a specific region
in one of the t-RNAs, m-RNAs, and r-RNAs (multitarget drugs). This
redundancy helps prevent the development of cancer cells resistant to
multiple chemotherapy treatments. Modifications that resist nucleases
also enable the fragments to remain in cancer cells and the bloodstream
longer.
Team Expertise: Our team uniquely integrates the
Theory of Inventive Problem Solving (TRIZ) into pharmaceutical
development and includes Ph.D. holders with diverse educational and
practical backgrounds. We are composed of experts in pharmacology,
microbiology, and drug design and have recently secured 10 new US
patents in drug design, including three in MDR, bringing our total to
over 220 granted patents, with several industry-implemented
innovations. Our principal investigator and consultant are the authors
of the proposal’s granted patent and a peer-reviewed published article.
Key personnel include a clinician experienced in anticancer trials and
multicenter studies. Our diverse expertise and systems approach
position us to tackle complex challenges effectively.
